
Apple, Johnson & Johnson Team Up on New Apple Watch Study
In an effort to accelerate the diagnosis of a leading cause of stroke, Johnson & Johnson and Apple have partnered on a new study that determines how the latest Apple Watch in conjunction with an app from the pharmaceutical company can help in effectively detecting atrial fibrillation, or AFib (via USA Today).
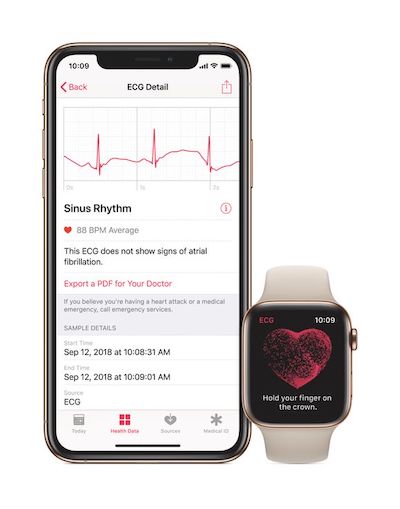
For those who don’t know, AFib is a common form of irregular heart rhythm that affects tens of millions of people and often goes undiagnosed until life-threatening complications occur. The condition is responsible for nearly 130,000 deaths and 750,000 hospitalizations each year in the U.S.
The latest Apple Watch Series 4 has an irregular heart rhythm notification feature, as well as an FDA-cleared ECG app, both of which are designed to detect AFib. According to Johnson & Johnson, the goal of the study is to show that there AFib is a lot more common in the real world than we ever imagined.
“We are receiving thank you letters daily from Apple Watch wearers who are discovering they have AFib,” said Apple Chief Operating Officer Jeff Williams. “We want a deeper understanding about outcomes and prevention associated with early detection. We are excited to work with Johnson & Johnson, which has a long history and expertise in cardiovascular disease.”
Paul Stoffels, Johnson & Johnson’s executive vice president and chief scientific officer, said “the goal is to identify early on AFib and prevent stroke by combining the physical know-how from Apple and what we have from the medical and scientific know-how.”
In case an AFib reading shows up, patients will be directed to seek a formal diagnosis from their medical provider.

